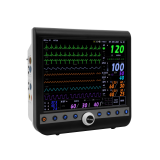
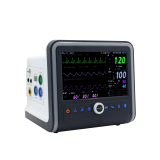
Modular type patient monitor, VM15std1
Votem's Modular monitor
Negotiable Min Order Quantity Unit
- Required Quantity
-
- Place of Origin
- South Korea
- Payment Terms
- T/T
- Production method
- Available
- Shipping / Lead Time
- Negotiable / Negotiable
- Category
- Multi-Parameter Monitor
| Product name | Modular type patient monitor, VM15std1 | Certification | - |
|---|---|---|---|
| Category | Multi-Parameter Monitor | Ingredients | - |
| Keyword | medical device , medical equipment , patient monitor , vital signs monitor | Unit Size | - |
| Brand name | - | Unit Weigh | - |
| origin | South Korea | Stock | 1 |
| Supply type | Available | HS code | 9018197000 |
Product Information
Modular Monitor VM15std1
│ DESCRIPTION │
Patient Monitor is an essential medical equipment analyzing the various vital signs of patient and showing waveform and numeric types of data to medical professionals so that they can apprehend the condition of patients.
│ CONFIGURATION │
- Standard : ECG, SpO2, NIBP, 1 TEMP
- Optional : EtCO2 (Respironics Mainstream or Sidestream),
MAIN Monitor Performances Specifications
l Display
n TFT Color LCD Display
n Waveform Display Method: Scroll
n Display Size: 15.0 inch
n Resolution: 1024 x 768 (XGA)
n Sweep Speed: 6.25, 12.5, 25, 50 mm/sec
n Display waveform Time: 2.5 sec
n Waveform Freeze: Function of waveform freeze
n Waveform Display: 3ch ECG, SpO2, 4ch IBP, Resp or EtCO2 or Multi-Gas, qCON
n Waveform Traces: 12 Waveforms Max.
n Measurement Value Display: Heart Rate, SpO2, Pulse Rate, NIBP, Resp, 4ch IBP, 4ch Temp, EtCO2 or Multi-Gas, ICO, qCON
l UI (User Interface)
n Touch screen: 15.0 inch Touch panel, 4-wire resistive type
n Encoder: Trim Knob
n Key buttons: Power, Alarm Snooze, Change Screen, NIBP start/stop, Print, Exit
n ICO (Cardiac Output)
n Oxy-CRG
l Sound
n Beep: OFF, 10~100%
n Alarm: 10~100% (Low / Middle / High)
n Information: OFF, 10~100%
l Languages
n English, French, German, Italian, Spanish, Portuguese, Portuguese-BR, Turkish,
Russian, Czech, Rumanian, Polish
TM (Transport Module) Performances Specifications
l Display
n TFT Color LCD Display
n Waveform Display Method: Scroll
n Display Size: 4.3 inch
n Resolution: 480 x 272
n Sweep Speed: 6.25, 12.5, 25, 50 mm/sec
n Display waveform Time: 2.5 sec
n Waveform Traces: 3 Waveforms Max.
l UI (User Interface)
n Key buttons: Power, Alarm Snooze, NIBP start/stop
l Parameters
n ECG
n Resp (Respiration)
n SpO2
n NIBP
n 1 Channel Temp
l Sound
n Beep, Alarm (Low / Middle / High), Information
n Volume: N.A (Fixed)
B2B Trade
| Price (FOB) | Negotiable | transportation | Express,Negotiation Other |
|---|---|---|---|
| MOQ | Negotiable | Leadtime | Negotiable |
| Payment Options | T/T | Shipping time | Negotiable |

- President
- Tony Kang
- Address
- Dongnae-myeon, Geodudanji 1-gil,27, Chuncheon-si, Gangwon-do, Korea
- Product Category
- Multi-Parameter Monitor,Oximeter
- Year Established
- 2007
- No. of Total Employees
- 1-50
- Company introduction
-
VOTEM is manufacturing Patient Monitor (for Human and Veterinary) and other medical products at own factory site, supplying these products to the countries in Europe, America, Asia, and other continents.
VOTEM has established in 2007 by research personnel with masters or higher degree in its related fields of study to create the new value in the medical device market.
VOTEM will focus all activities to satisfy the needs of the customers through the value innovation. Also, VOTEM is planning to lead the medical industry by carrying out multiple research projetcts and clinical studies, establish and maintain the quality system in accordance with ISO13485 and Medical directive, 93/42/EEC.
- Main Markets
-
 Australia
Australia
 Malaysia
Malaysia
 Philippines
Philippines
 Romania
Romania
 U.A.E.
U.A.E.
- Main Product



































 South Korea
South Korea






_2.jpg)
